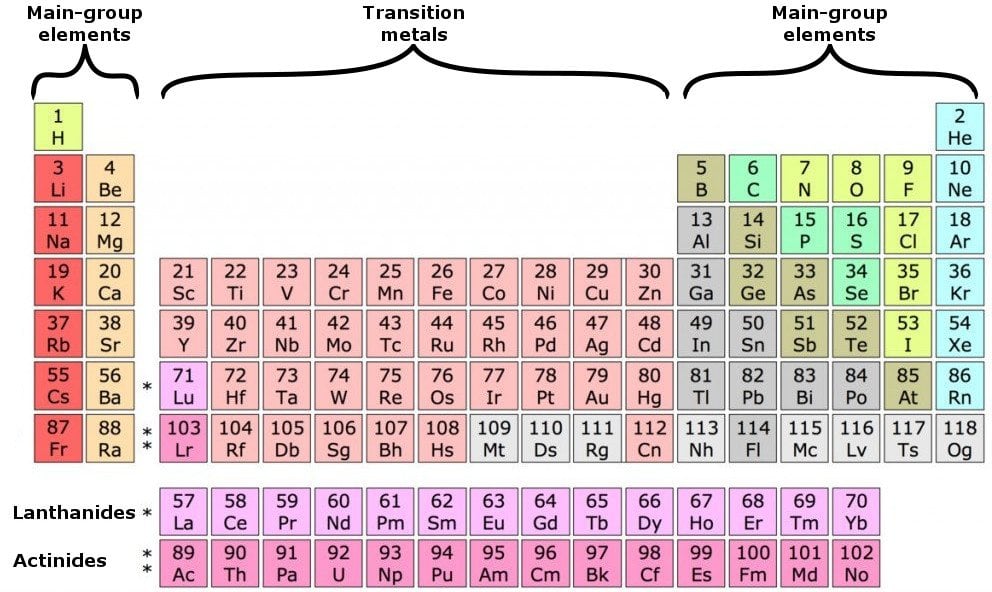
Okay, so for chlorine, we see that we have our seven valence electrons in the outer shell, with all the remaining electrons serving as our inner core electrons. And in this third show we have our seven valence electrons, The remaining 10 electrons are in shells too, And one they total up to a total of electrons. And you can see here with this representation of the chlorine atom We have here are 3rd shell, so and equals three. has a smaller atomic radius the sodium, which means the valence electrons are closer to nucleus, therefore require more energy to remove an electron when. It explains how to determine the number of valenc. The position of an atom in the periodic table represents the number of valence electrons present in the outermost shell of the atom. So that would mean out of the 17 total electrons. This chemistry video tutorial provides a basic introduction into valence electrons and the periodic table. Chlorine has an atomic number of 17 When it is neutral it has 17 protons. So you're gonna say total electrons Which is connected to your atomic number for neutral element equals your valence electrons plus your inner core electrons. How to determine the number of valence electrons and draw Lewis structures for main group elements starting from the electron configuration. 4:26, Sal says 'In most cases, your valence electrons are going to be your outermost electrons. 
We're going to say here that your total number of electrons. The number of valence electrons in an atom governs its bonding behavior. Now, for the sake of this video, I'm gonna focus most on the extremes of the periodic table, the groups at the left and the right. So these are the remaining electrons that are not valence electrons. And because elements with similar valence electrons will have similar reactivities, they will form similar ions. Now, besides the valence electrons, all the remaining electrons are called your inner core electrons. Now these valence electrons are the outer shell electrons involved in forming chemical bonds. Now for main group elements were going to say that the number of valence electrons that they possess equals their group number.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
